
Many of the necessary processes for bio-fuels production are related to the upgrade of the oxygen-rich hydrocarbons to make them better transportation fuels (see here and here). The high level of oxygen content is even an issue with our most voluminous biofuel ethanol, which is 34% oxygen by weight. What if you could develop a biological process that yielded fuels that didn't need to be upgraded? Fuels that were already devoid of oxygen moieties? That is the current work that is going on at Calysta Energy, as reported by their CTO Dr. Josh Silverman.
The challenges
In addition to the inferior fuel quality of oxygenated hydrocarbons, using them going uphill - from more to less oxidized - is not economical, as excellently presented by the Spring Meeting's opening keynote speaker, William Bernholzer (see this post and this interview). If sugars and vegetable oils are roughly cost equivalent to petroleum on a carbon basis, how is a process of bioconversion of them to fuel that has a 35% theoretical (and 20-25% actual) yield ever going to be economical? The optimal approach would be to use the abundant methane as a feedstock and build higher molecular weight hydrocarbons from that. Methane is also the cheapest carbon source available (aside from coal).
The first challenge then is to find and/or modify an organism that can use methane as both its primary energy source and also a feedstock for products fermentation. Calysta Energy has been working with proprietary cells that can live off of only methane as carbon and energy source, plus some inconsequential amounts of nutrient salts and trace metals. There is a roughly 50:50 split between conversion and energy use of the methane, and it is a very energetic process (52 mega-joules per Kg of cell mass), so heat transfer is a critical design element. The very low solubility of methane in the aqueous growth media is also a challenge to reactor design. Several designs have been evaluated and are detailed next.
Reactor types
- Traditional Stirred Tank Fermenter This design is a high aspect ratio (H/D) vertical mixed tank with gas sparged under the turbine agitator. These time-tested designs are most useful when the primary reactant is in the liquid phase, or a solute, as the mass transfer rates from the vapor phase are low, on the order of a KLa of 200/hr*.
- Air-Lift Fermenter In this design, the mixing is provided by the movement of liquid and gas up a central cylinder with the liquids returning in down-flow in the annular spaces. Interphase mass transfer is improved, though there is great variability from the bottom (high rates) to the top (low rates) of the central tube. Heat transfer is also problematic as the mass of of the down-flowing liquids hinders transfer from the central core.
- U-Loop Fermenter In this unique Scandinavian design, gas is injected at the TOP of a reactor tube where the liquid phase is recirculating downward at a velocity somewhat faster than the rise-rate of the gas bubbles. Control of the two phases' flow rates allows control of the contact or residence times and hence conversions, with a KLa on the order of 1,200/hr. As the bubbles move downward, the loss of mass (from transfer) and the static pressure increase reduces the bubble size, thus increasing the area to volume ratio. In addition, the design allows for excellent heat transfer, and minimal head space volumes of the explosive methane-oxygen mixture.
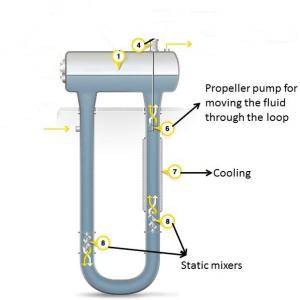
- Concentric U-Loop Design- One final system under consideration is one which is in concept a U-Loop Fermenter rotated through 360? to give a hybrid of the U-Loop (from a flow perspective) and the Air-Lift Fermenter (from a geometric standpoint). It has many of the benefits of each, but also some of the drawbacks.
* KLa is a critical measure of fermenter design efficiency. It is the product of the mass transfer coefficient (gas to liquid phase) and the interfacial area per unit volume, with units of hour-1.
Progress and plans
With conversion yields 2x higher and a unit carbon cost 1/5 of sugars, methane holds an order of magnitude advantage in economics as a source for biofuels and materials. Calysta Energy is currently working with Nature Works on production of lactic acid from methane using these technologies. Other concepts that are under study include coating the organisms on flat surfaces to create "plate and frame" bio reactors, and in collaboration with the Lawrence Livermore Labs evaluating using nanostructures as the support for the organisms.

For more information, contact Dr. Josh Silverman, CTO Calysta Energy at jsilverman@calystaenergy.com


