
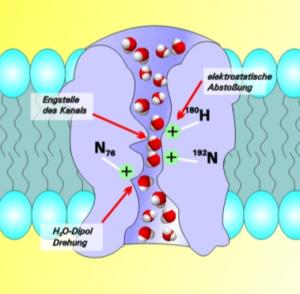
Aquaporin
(AQP) is an hour-glass shaped protein found in cell membranes that has a unique functionality and properties of interest in water transport, including:- Size restriction of molecular transport, of about 2.8 Angstroms
- Positively charged amino acid regions that effectively reject cation passage, and
- Water dipole reorientation functionality that essentially guarantees only water "makes it through."
Efforts are underway at the National University of Singapore to better understand these proteins, and how to incorporate them into engineered structures. Presentations of some of the current research on AQP utilization were made Monday by Guo Fei Sun and Phuoc H. H. Duong.
Incorporating AQP into a lipid bi-layer

The particular variety of AQP currently under study is AQP-Z, which is harvested from E. coli bacteria. In order to maintain protein stability and functionality, a detergent is used in solution to create a protective micelle. Once protected by this carrier, the AQP-Z can be incorporated into synthetic lipid liposome structures, thus providing a controlled path for water from outside to enter the liposome (or vice versa). Once the AQP-Z is incorporated, the free detergent must be removed using biobeads. Free surfactants will interfere with the operation of the AQP-Z.
Langmuir-Blodgett bi-layer techniques
To create bi-layer films, a variation of the Langmuir-Blodgett method of creating mono-layer films can be used. Again, utilizing a detergent as a "carrier," the AQP-Z is incorporated it into a DPPC-DOGSNTA mono-layer, the former being the main mono-layer constituent, and the latter the AQP-Z "catcher" molecule.
Once incorporated and detergents removed, the "seeded" mono-layer is combined with a "pure" monolayer to form a bi-layer with operational AQP available to transport water from one side to the other. Each AQP-Z "channel" can transport up to 3 billion molecules/second across the film.
A more permanent membrane

Supporting such an AQP enhanced bi-layer on a porous substrate could potentially allow said system to function in a desalination capacity.
To make such systems more structurally stable, block co-polymers with properties similar to lipids are utilized. Polymers with an A-B-A structure, with the A blocks being hydrophilic and the B block hydrophobic, thus act just like dipoles in water, and can form vesicle structures as well as mono and bi-layers suitable to incorporate the AQP-Z. One AQP-Z per 50 polymer molecules was shown to give optimal water transport.
Typically, a porous alumina substrate has been used to support the engineered AQP-Z film. The hydrophilic polymer ends contain a disulfide moiety to facilitate attachment to the substrate. Preliminary testing of the supported films yields a salt retention of 30-50% at 75 psi. Extensive research is ongoing at NUS to further characterize and optimize these processes.


